Event date:
Sep
17
2021
4:00 pm
Development of Silica supported catalysts for Depolymerization of Lignocellulosic Biomass via Lignin-first approach
Supervisor
Dr. Muhammad Zaheer
Student
Noor Ul Huda
Venue
Zoom Meetings (Online)
Event
PhD Synopsis defense
Abstract
About 95% of the carbon-containing chemicals that sustain our daily lives are obtained from crude oil. Rapid depletion of crude oil supplies and escalating environmental effects of fossil resource utilization have led to the current strong drive, widespread interest, and investments in renewable resources to synthesize chemicals. Biomass is the only renewable carbon resource to derive both fuels and chemicals among various available substitutes. Lignocellulose is the most abundant form and, being inedible biomass, will not impose a direct negative impact on food supplies. Therefore, it serves as an ideal feedstock for replacing fossil resources in producing functional chemicals.
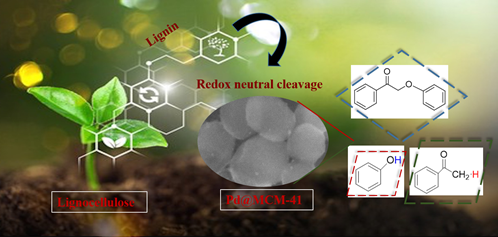
Lignocellulosic biomass primarily consists of lignin and carbohydrates (cellulose and hemicellulose). The latter has been extensively studied and observed to be a versatile feedstock for the production of fuels and chemicals. However, the efficacy of lignin—the largest source of natural aromatics—is still untapped due to its high recalcitrant nature and complex structure. Efficient lignin depolymerization with an effective catalytic system followed by targeted upgrading could increase the environmental sustainability and economic viability of chemicals. In this regard, the rapid evolution of nanomaterials has made breakthroughs to design and synthesize high-performance metal-based catalysts for novel lignin conversion technologies. However, generating highly dispersed metal nanoparticles (NPs) of the desired size without aggregation is an important challenge for enhancing the catalytic activity of supported catalysts. It has been found that a strong interaction between the metal precursor and the support during catalyst synthesis ensures the formation of monosized and uniformly distributed NPs.
We envisage using transition metal complexes in place of metal salts to better interact the metal precursor with the support. It would also avoid metal loss due to the precipitation of metal hydroxides during basic reaction conditions. In an initial attempt, we successfully synthesized well-dispersed palladium (Pd) NPs in mesoporous MCM-41 matrix by the thermal decomposition of MCM-41 loaded with a metal β-diketone complex under various gaseous atmospheres. Complete characterization of the catalyst included X-ray Powder Diffraction (PXRD), Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM), X-ray Photoelectron Spectroscopy (XPS), and nitrogen physisorption (BET).
The catalyst calcined under air showed the complete redox neutral cleavage of a lignin model compound without requiring any external hydrogen source. The catalyst showed a 99% conversion of 2-phenoxy-1-phenyl-ethanol to 2-phenoxy-1-phenyl-ethanon a 90 ℃ under an inert atmosphere and produced 75% 2-phenoxy-1-phenyl-ethanon and 16% and 9% yields of acetophenone and phenol, respectively.
Lignocellulosic biomass primarily consists of lignin and carbohydrates (cellulose and hemicellulose). The latter has been extensively studied and observed to be a versatile feedstock for the production of fuels and chemicals. However, the efficacy of lignin—the largest source of natural aromatics—is still untapped due to its high recalcitrant nature and complex structure. Efficient lignin depolymerization with an effective catalytic system followed by targeted upgrading could increase the environmental sustainability and economic viability of chemicals. In this regard, the rapid evolution of nanomaterials has made breakthroughs to design and synthesize high-performance metal-based catalysts for novel lignin conversion technologies. However, generating highly dispersed metal nanoparticles (NPs) of the desired size without aggregation is an important challenge for enhancing the catalytic activity of supported catalysts. It has been found that a strong interaction between the metal precursor and the support during catalyst synthesis ensures the formation of monosized and uniformly distributed NPs.
We envisage using transition metal complexes in place of metal salts to better interact the metal precursor with the support. It would also avoid metal loss due to the precipitation of metal hydroxides during basic reaction conditions. In an initial attempt, we successfully synthesized well-dispersed palladium (Pd) NPs in mesoporous MCM-41 matrix by the thermal decomposition of MCM-41 loaded with a metal β-diketone complex under various gaseous atmospheres. Complete characterization of the catalyst included X-ray Powder Diffraction (PXRD), Scanning Electron Microscopy (SEM), Transmission Electron Microscopy (TEM), X-ray Photoelectron Spectroscopy (XPS), and nitrogen physisorption (BET).
The catalyst calcined under air showed the complete redox neutral cleavage of a lignin model compound without requiring any external hydrogen source. The catalyst showed a 99% conversion of 2-phenoxy-1-phenyl-ethanol to 2-phenoxy-1-phenyl-ethanon a 90 ℃ under an inert atmosphere and produced 75% 2-phenoxy-1-phenyl-ethanon and 16% and 9% yields of acetophenone and phenol, respectively.
Meeting Link: https://lums-edu-pk.zoom.us/j/91813465320?pwd=WUlDQW5reENkOTNZeURNbXhtSEo5UT09
Meeting ID: 918 1346 5320
Passcode: 246830

